 
IMMEDIATELY transport the victim to a hospital. If the victim is convulsing or unconscious, do not give anything by mouth, ensure that the victim's airway is open and lay the victim on his/her side with the head lower than the body. Be prepared to transport the victim to a hospital if advised by a physician. If the victim is conscious and not convulsing, give 1 or 2 glasses of water to dilute the chemical and IMMEDIATELY call a hospital or poison control center. Whenever possible, Self-Contained Breathing Apparatus (SCBA) should be used if not available, use a level of protection greater than or equal to that advised under Protective Clothing. Provide proper respiratory protection to rescuers entering an unknown atmosphere. If symptoms (such as wheezing, coughing, shortness of breath, or burning in the mouth, throat, or chest) develop, call a physician and be prepared to transport the victim to a hospital. INHALATION: IMMEDIATELY leave the contaminated area take deep breaths of fresh air. If symptoms such as redness or irritation develop, IMMEDIATELY call a physician and be prepared to transport the victim to a hospital for treatment. Gently wash all affected skin areas thoroughly with soap and water. SKIN: IMMEDIATELY flood affected skin with water while removing and isolating all contaminated clothing. IMMEDIATELY transport the victim after flushing eyes to a hospital even if no symptoms (such as redness or irritation) develop. Do not put any ointments, oils, or medication in the victim's eyes without specific instructions from a physician. Flush victim's eyes with water or normal saline solution for 20 to 30 minutes while simultaneously calling a hospital or poison control center. Normalized Breakthrough Times (in Minutes) ChemicalĮYES: First check the victim for contact lenses and remove if present. Information of DuPont or others covering any material or its use. Recommendation to infringe any patent, trademark or technical This information is not intended as a license to operate under or a Warranties of merchantability or fitness for a particular use andĪssume no liability in connection with any use of this information. Warranties, express or implied, including, without limitation, no Since conditions of use are outside our control, DuPont makes no are damaged, end user shouldĭiscontinue use of garment to avoid potential exposure to chemical. Ifįabric becomes torn,abraded or punctured, or if seams or closures fail, In many cases, seams and closures have shorterīreakthrough times and higher permeation rates than the fabric. Information should first verify that the garment selected is suitableįor the intended use. 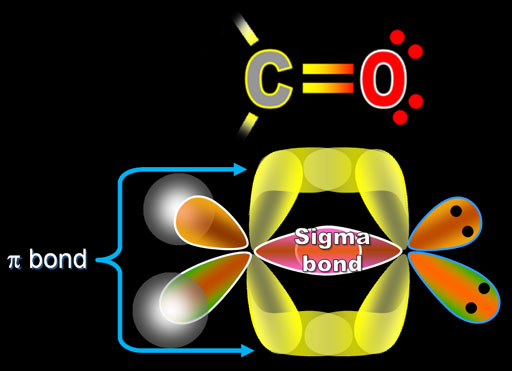
Responsibility to determine the level of toxicity and the proper Having technical skill for evaluation under their specific end-useĬonditions, at their own discretion and risk. It is intended for informational use by persons Laboratory performance of fabrics, not complete garments, underĬontrolled conditions. 
Soc.This information is based upon technical data that DuPont believes toīe reliable on the date issued. There is no more than 1 sigma bond between any two atoms. In the polyacenes, the polyeneyl orbitals interact in a pi-sense, while in the compounds we happened upon (which we now call "sigma-polyacenes"), the polyenyl orbitals interact face-to-face, in a sigma-sense, as shown above (orbital cartoons are shown at the left and the actual HOMO and LUMO from our calculations are shown at the right). The anthracene molecular formula is C14H10, it has three rings so that the rule contributes the number of sigma bonds as 24 added with 3 is 27, which will be subtracted with 1 which will give 26. in that both involve the interaction of polyenyl chains. These structures are similar to a class of molecules called "polyacenes" - ribbons of fused aromatic rings like anthracene, tetracene, etc. While MO theory deals with sigma bonds as well as pi bonds, we will restrict. When we explored discrete model systems for this polymer, we discovered that, rather than structures with localized sigma-bonds connecting their two polyene chains, structures were formed that appeared to have two polyenyl radical chains interacting with each other through space (see the structure above for an example, selected distances are shown in angstroms). We had hoped that polymers of this type would undergo degenerate Cope rearrangements ( sigmatropic shifts) with low activation barriers, making for fluxional polymers that could transport the electrons in a sigma-bond back-and-forth along their backbones of parallel pi-bonds. One of our initial proposals for a potential " sigmatropic shiftamer" is shown above. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
